Thermal shock is common in many industries, including manufacturing, metallurgy, ceramics, and glass production. When a material or machine suffers thermal shock, its integrity is compromised, posing safety risks and further product failure.
Thermal shock refers to the mechanical failure of a material due to a rapid and significant change in temperature. This phenomenon can result in the material becoming cracked, fractured, or otherwise compromised. As such, many industries must implement measures to prevent it during production.
The primary cause of thermal shock is the uneven expansion or contraction of a material’s components when exposed to rapid temperature changes. In most materials, thermal shock is undetectable unless the material is quality-tested. However, it’s easier to identify in glass, as glass surfaces are particularly vulnerable to thermal shock.
Everyday Illustration of Thermal Shock
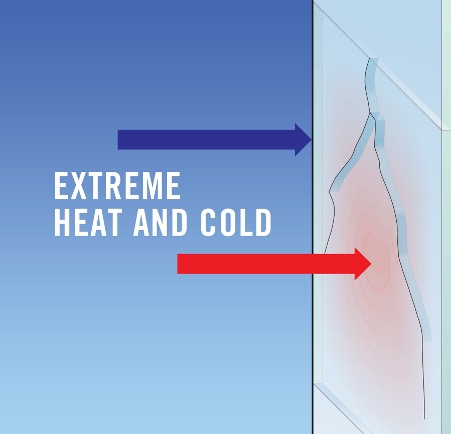
Most of us use glass in some way in our daily lives. It’s in our windows, our vehicles, lights, and especially in our kitchens. Given the prevalence of glass around us, you’re likely to witness thermal shock at play. For example, compromised windows and windscreens can develop cracks overnight in locations with extreme air temperature fluctuations.
Additionally, fine kitchen glassware is particularly susceptible to thermal shock. For instance, wine glasses can easily crack if exposed to rapid temperature changes, e.g., during washing. Placing a delicate wine glass in hot water will likely lead to cracking if the temperature change is too significant and fast.
Thermal Shock in the Industrial World

Thermal shock poses a significant concern in the industrial world, as it can have far-reaching effects. For instance, it can compromise machinery, products, services, and human safety if not prevented. It can also result in significant losses due to failure and wastage. Therefore, industries need to understand, manage, and prevent thermal shock as far as possible.
Let’s look at the use of glass in the food and beverage industry as an example:
In Secomak’s experience, the most common example of thermal shock in glass is the cracking of food and beverage glass bottles and jars during the hot filling process. Typically, empty containers are stored on pallets at ambient temperatures as low as 5°C before being filled. When these receptacles are filled with cooked products in excess of 90°C, they risk cracking due to thermal shock.
In such instances, the weakest point of the jar or bottle is susceptible to cracking, i.e., the bottleneck where the glass is thinnest.
The Risk of Thermal Shock: Understanding Delta T

In industrial heating systems, Delta T or ∆T refers to the temperature difference between the input and output of a heating system, heat exchanger, or material. ∆T is often used in discussions related to the efficiency and performance of heating systems, where a higher Delta T indicates heating efficiency.
The equation to calculate Delta T is as follows:
∆T = T1 – T2
T1 refers to the input (starting) temperature, while T2 is the output or new temperature.
While preferable for measuring heating efficiency, a high Delta T is not ideal for applications where the materials are prone to thermal shock. The extreme and rapid temperature changes can cause differential expansion, leading to internal stress and cracking or failure.
To illustrate, ∆T for the bottles that cracked during filling is calculated as follows:
∆T = T1 – T2 = 5°C – 90°C = -85°C
It’s recommended that ∆T should not exceed 35°C when considering the surface temperature of glass and the contents with which it is filled.
This means the glass bottles were abruptly exposed to a temperature contrast of 85°C, leading to thermal shock at their weakest point. Secomak’s solution to mitigate thermal shock in this application was to preheat the bottles to approximately 60°C, thereby minimising Delta T from the initial 85°C to <35°C.
Mitigating Thermal Shock: Heating Strategies
Industries can mitigate thermal shock in several ways, including the following:
- Ensuring a gradual temperature change for the object or material,
- Avoiding temperature gradients within the material by providing even heating,
- Using thermal barriers (like insulation),
- Choosing materials more resistant to thermal shock,
- Considering the product design concerning the manufacturing process and
- Preheating and precooling materials or objects.
An excellent way to mitigate thermal shock in applications such as the food and beverage industry is by preheating receptacles before filling them with hot contents. There are a few ways to do this, and the chosen method will depend on the application.
However, steam and infrared heating are two standard preheating methods used in this industry.
Steam Heating
Steam heating is an excellent way to preheat materials before filling them with hot contents. In such applications, the cans or bottles are fed through a steam tunnel using a conveyor system. As the objects move through the small industrial sauna, they are uniformly heated and sterilised. Then, as they pass out of the tunnel, they are air-dried in a drying machine before introducing hot contents.
The Pros of Steam Heating
Steam heating objects is ideal when a whole container must be uniformly heated, as the steam within the tunnel envelopes the entire object. It is an effective heating method as it is non-abrasive, easily controlled, and sterilises and cleans the object before filling.
The Cons of Steam Heating
Steam tunnels, while effective, are costly and have a large footprint. Given the presence of moisture, steam tunnels must be made with stainless steel, which is pricey. Additionally, the process wastes filtered water and energy to create steam.
Finally, suppose the objects aren’t sufficiently dried. In that case, the residual moisture can damage the ingredients or product labels or leave undesirable watermarks. Hence, it becomes necessary to use a specialised drying machine.
Infrared Electrical Heating

Infrared heating helps mitigate thermal shock in localised areas of an object or material, e.g., bottlenecks. It is more efficient than creating an entire heating chamber and doesn’t require a water-resistant tunnel or additional blowers.
The process of heating objects using infrared is more straightforward than steam heating. For example, suppose it’s only the bottlenecks that must be heated. In that case, the infrared lights will be positioned optimally within the heating tunnel. As the bottles pass through on the conveyor belt, the heat will only warm the applicable part of the bottle before it is filled with warm contents.
The Pros of Infrared Heating
Since infrared heating is localised, it is a much more cost-effective way of preheating an object or material. It is helpful in applications where the contents of a prefilled bottle are heat-sensitive, and steam heating is not ideal.
The Cons of Infrared Heating
Infrared preheating is only efficient for heating small, localised areas of an object or material. It can be used for more extensive applications but would require more strategically placed lights and electricity to power them.
Conclusion
Thermal shock threatens the integrity of some manufacturing materials, so industries must understand and implement ways to prevent it altogether. To minimise Delta T, an efficient means to mitigate thermal shock is to preheat objects during the production process.
Secomak offers excellent preheating solutions for the food and beverage industry. Our safe and effective bottle and can drying solutions utilise steam or infrared to preheat and dry containers before filling them with hot contents.



